OXIDATION AND REDUCTION
Redox reaction include all chemical reaction in which atoms have their oxidation state changed in general redox reaction involved the transfer of electron between species, Oxidation is the lost of electron or an increased in oxidation state by a molecule, atom, or ion. Reduction is the gain of electron or a decrease in oxidation state by a molecule, atom or ion.
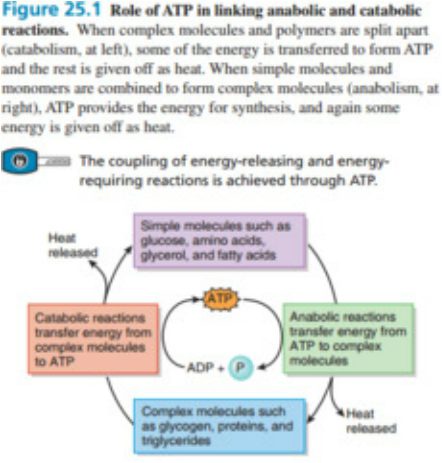
ROLE OF ATP
Adenosine triphosphate (ATP) is the energy currency for cellular processes provides the energy for both energy-consuming endergonic reactions and energy-releasing exergonic reactions. When the chemical bonds within ATP broken, energy is released and can harnessed the cellular work. The more bonds in a molecule shows the more potential energy contains. Because the bond in ATP is so easily broken and reformed, like a rechargeable battery that powers cellular process from DNA replication to protein synthesis. The two bonds phosphoanhydride bonds between the phosphates are equal high-energy bonds, when broken it release sufficient energy to power a variety of cellular reactions and processes. ATP breakdown into ADP and Pi is called hydrolysis because it consumes water molecule from (hydro-, meaning "water", and lysis, meaning "separation").