WATER CHEMISTRY
Water is the most abundant compound on the Earth's surface. In nature, water exists in the liquid, solid, and gaseous states. It is in dynamic equilibrium between the liquid and gas states at 0 degrees Celsius and 1 atm of pressure. At room temperature (approximately 25 degrees Celsius), it is tasteless, odorless, and colorless liquid. Many substances dissolve in water, and is commonly referred to as the universal solvent
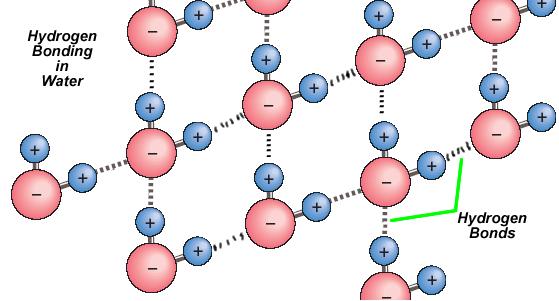
The polar nature of water is a particularly important feature that contributes to the uniqueness of substance. The water molecule forms angle with oxygen atom at the vertex and hydrogen atoms at the tips. Because oxygen has a higher electronegativity than hydrogen, side of the molecule with the oxygen atom has a partial negative charge. An object with such a charge difference is called a dipole (meaning "two poles"). The oxygen end partially negative, and the hydrogen end is partially positive; because of this, the direction of the dipole moment points from the oxygen toward the center position between the two hydrogens. This charge difference causes water molecules to attracted to each other (the relatively positive areas are attracted to the relatively negative areas), as well as to other polar molecules. This attraction contributes to a hydrogen bonding and explains many of water's properties.
Water molecules are amphiprotic and can function both acids and bases. One water molecule (acting as a base) can accept hydrogen ion from a second one (acting as an acid). This will be happening anywhere when there is even a trace of water it does not have to be pure. A hydronium ion and a hydroxide ion are formed.
The polar nature of water is a particularly important feature that contributes to the uniqueness of substance. The water molecule forms angle with oxygen atom at the vertex and hydrogen atoms at the tips. Because oxygen has a higher electronegativity than hydrogen, side of the molecule with the oxygen atom has a partial negative charge. An object with such a charge difference is called a dipole (meaning "two poles"). The oxygen end partially negative, and the hydrogen end is partially positive; because of this, the direction of the dipole moment points from the oxygen toward the center position between the two hydrogens. This charge difference causes water molecules to attracted to each other (the relatively positive areas are attracted to the relatively negative areas), as well as to other polar molecules. This attraction contributes to a hydrogen bonding and explains many of water's properties.
Water molecules are amphiprotic and can function both acids and bases. One water molecule (acting as a base) can accept hydrogen ion from a second one (acting as an acid). This will be happening anywhere when there is even a trace of water it does not have to be pure. A hydronium ion and a hydroxide ion are formed.